
MEET SUSTAINABILITY GOALS
Sustainability data captured and centralised into the Hub dashboard includes: Scope 3 Emissions (Purchased goods and services, business travel, employee commuting, waste disposal, use of sold products), transportation and distribution (up- and downstream), investments and leased assets and franchises.
Integrated emissions calculations based on industry best practice, the GLEC framework and compliant with ISO 14083.
Track products along the value chain: reducing inventory safety buffers and respond quickly to unexpected events.
High quality analytics and reporting to achieve better planning, visibility, diagnostics and troubleshooting.
Contact Veratrak
Regulations are evolving to facilitate sustainable supply chain operations
The world has six years of carbon budget left to limit global temperature increase to 1.5°C compared to pre-industrial levels, according to the Mercator research Institute on Global Commons and Climate Change, and taking action to achieve tangible results is now more critical than ever before.
Healthcare stakeholders must play their part by accelerating the delivery of patient-centric, equitable, net zero health systems.
In our latest edition white paper, we take a look at some of the key environmental reporting frameworks and standards.
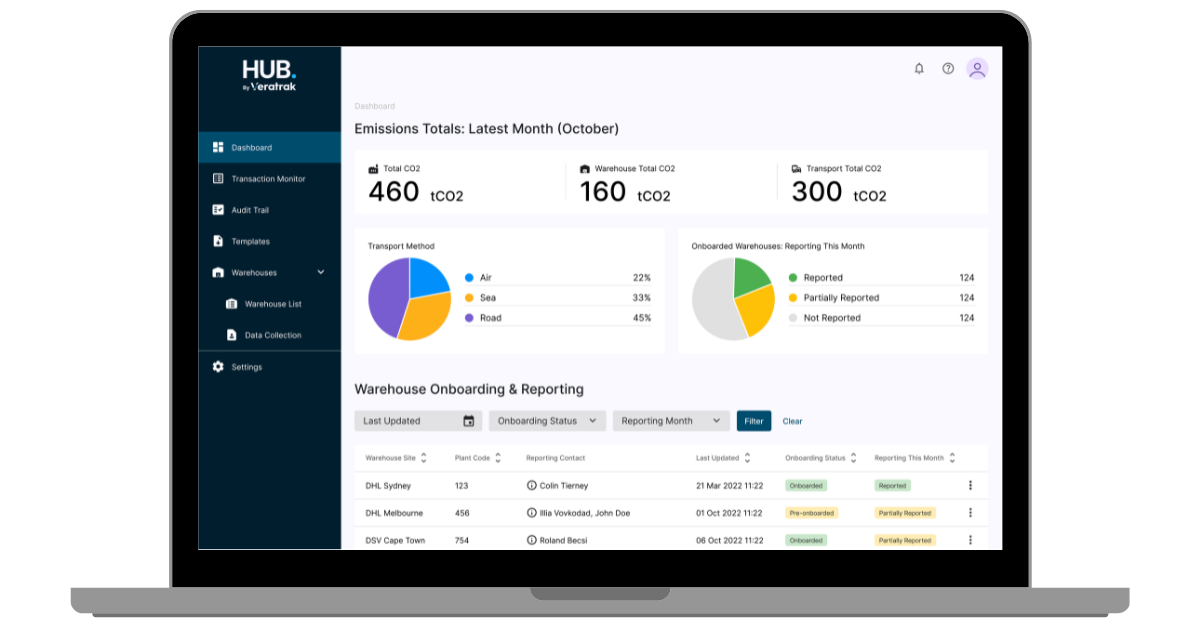
All your suppliers' data managed from a single dashboard
The Hub integrates directly with your supply chain systems, so you can manage your data in real time from a unified Hub dashboard.
Achieve better visibility on logistics provider activities and shipping workflows
Capture packaging requirements from delivery method, timelines, currency, to warehousing
Upgrade your IT infrastructure seamlessly and reduce system migration risks
Safeguarding Data Integrity
Veratrak's solution architecture secures data ingested to the Hub through a private blockchain network, ensuring leading security and compliance with GxP Data Integrity, FDA 21 CFR Part 11 and EU GMP.

.png)


